 
Ward's® Periodic Table of the Elements Flash CardsĮach card contains the atomic symbol, the element name, atomic number, atomic weight, electron configuration, period, and element family.  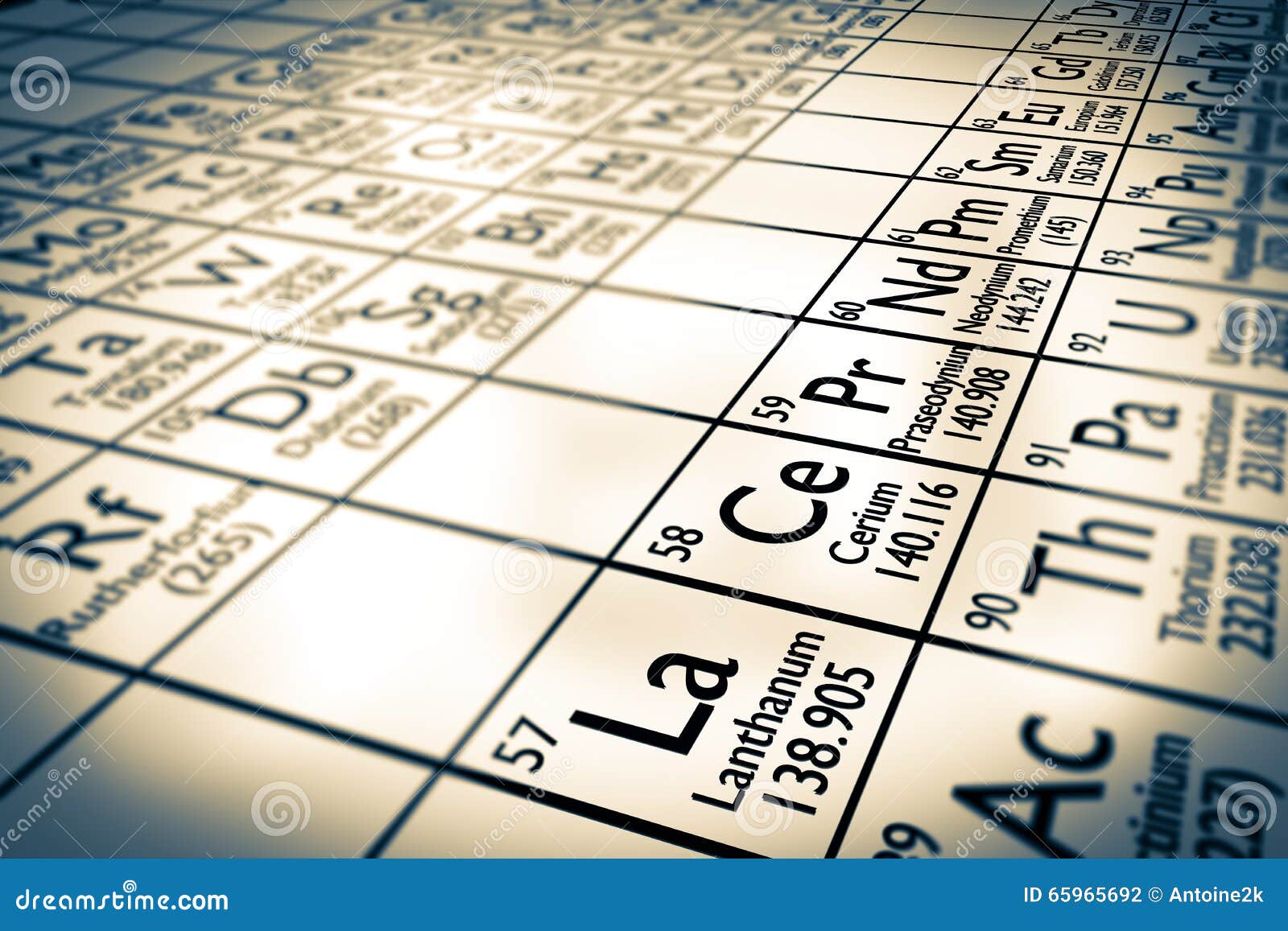
Everything we create from computer motherboards, towering skyscrapers, bridges, medicine, clothing, and everything else comes from our ability to harness the properties of the elements for our benefit. Sodium - Although abundant on Earth, sodium is never naturally found in its pure form it forms compounds with other elements.Įlements are the building blocks of life and human innovation.Oxygen - Oxygen is the third most abundant element in the universe however, its reactivity made it relatively rare in Earth's early atmosphere.Neon is the 5th most common element in the universe but comprises only 0.0018% of the Earth's atmosphere. It doesn’t respond with oxygen, helium, neon, and argon. Fluorine - Fluorine is the most receptive and electronegative of all the chemical elements.The iron and steel industries depend on it. charcoal from wood and coke from coal) is used in metal smelting. Carbon - The amount of carbon on Earth is relatively constant it’s simply transformed from one form to another by the carbon cycle.Lithium chloride is one of the most hygroscopic (absorbs moisture from the air) materials known and is used in air conditioning and industrial drying systems (as is lithium bromide). Lithium - Lithium is the lightest of all the metals and can easily float on water.Hydrogen can be combined with carbon dioxide to make methanol or dimethyl ether (DME) which are important transport fuels. Hydrogen -Although it’s rare on Earth, Hydrogen is the most common element in the universe.Helium - Helium atoms are so light that they can escape Earth's gravity! Helium is used in rocket propulsion (to pressurize fuel tanks, especially those for liquid hydrogen, because only helium is still a gas at liquid-hydrogen temperature).Beryllium ceramics are used to focus and control eye surgery lasers. Beryllium - Beryllium is two-thirds the density of aluminum and has six times the specific stiffness of steel by weight.Here are just a few that your students will enjoy learning: There are so many fun facts to discover about the different elements. But those just scratch the surface of the 118 elements. Of course, they may be familiar with some elements like glittering gold, iron, oxygen, and the voice-altering helium. Your lesson plans help students discover how the things they see every day (including the face in the mirror) contain these common and unusual elements. (In the modern periodic table, a group or family corresponds to one vertical column.Do the budding chemists in your classroom have questions about the stuff that everything is made of? Their curiosity is understandable since everything in nature, from the clouds to the trees and the air we breathe, is made of elements. The periodic table allows chemists a shortcut by arranging typical elements according to their properties and putting the others into groups or families with similar chemical characteristics. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 118 known elements. The term “periodic” is based on the discovery that elements show patterns in their chemical properties at certain regular intervals. Mendeleev left spaces for elements he expected to be discovered, and today’s periodic table contains 118 elements, starting with hydrogen and ending with oganesson, a chemical element first synthesized in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, by a team of Russian and American scientists. Its story is over 200 years old, and throughout its history, it has been a subject for debate, dispute and alteration.Īttempts to classify elements and group them in ways that explained their behavior date back to the 1700s, but the first actual periodic table is generally credited to Dmitri Ivanovich Mendeleev, a Russian chemist who in 1869 arranged 63 known elements according to their increasing atomic weight. Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
